In regulatory affairs, a dossier is a term used to describe a compilation and array of documents regarding the safety, efficacy, and quality information of a medical product. The dossier is to be submitted to a regulatory authority for review, and approval of the submission would grant the product marketing authorization.
The compilation of the dossier has to follow certain formats and requirements. The International Council for Harmonisation (ICH) has published requirements for compiling technical documents of a medical product. This document is called the Common Technical Document (CTD). Many of the ICH member countries (which includes many European countries, the United States, Canada, Japan, China, etc.) are adopting the CTD in drug submissions to eliminate the need to reformat the information for submission to various different countries.
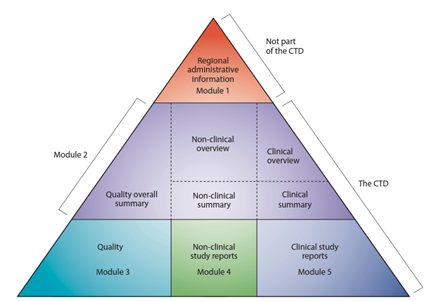
The CTD is composed of 5 modules, with each module containing specific information. Module 1 is technically not part of the CTD because this part is not harmonized and is region-specific. For example, the FDA has a different requirement for how Module 1 should be organized and presented in comparison to those submitted in European countries. In general, Module 1 contains administrative information, such as cover letters, application forms, contact information, patent and exclusivity information, references, meetings, etc. Remember, each region has different requirements as to what types of administrative information should be included and how they should be organized.
Modules 2, 3, 4, and 5 are harmonized sections for all regions. This means that ICH countries must follow the same format for these modules. Since 2003, the CTD has become the mandatory format for new drug applications in the EU, Japan, Canada, and the United States. Module 2 is a summary section containing an overall summary of the non-clinical, clinical, and quality information. Module 3 details the overall quality information and is also the section for building quality in the manufacturing process. This section contains the Chemistry Manufacturing and Controls (CMC) of the medical product. Module 4 is a section detailing the overall non-clinical information such as pharmacodynamics, drug interactions, pharmacokinetics in terms of absorption, distribution, metabolism, excretion, toxicology, and study reports. Module 5 details the overall clinical information such as biopharmaceutics, clinical pharmacology, efficacy, safety, benefits and risk conclusion, and study reports. Refer to the Figure below for an illustration of the CTD triangle.
Many regulatory authorities today are mandating the submission of CTD electronically which has led to the development of eCTD guidance and requirements. The eCTD is an electronic version of the CTD. Instead of submitting paper documents, the submissions are being submitted through a secured electronic portal.
For more information on how to submit a dossier or need help compiling one, please contact us for details.