On March 6, 2023, the Food and Drug Administration (FDA) launched a new directory of ingredients used in dietary supplements. The directory will be a “one-stop shop” for ingredient information that was previously found on different FDA web pages. The purpose of the new directory is to help manufacturers, retailers, and consumers make informed health by providing quick access to information about the ingredients which may be found in dietary supplements.
Within the new ingredient directory are links to 27 ingredients which can be accessed here.
Here’s an example of an ingredient as it appears in the FDA’s ingredient directory:
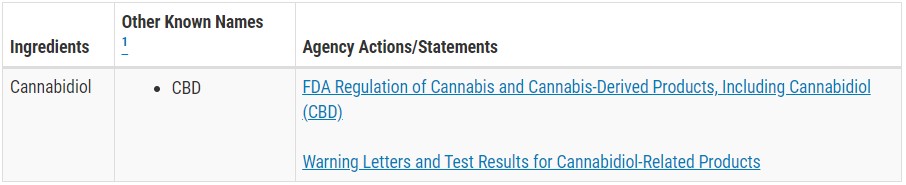
The FDA has stated that the directory is “not intended to be a comprehensive list of all ingredients” used in dietary supplements, and may not include all actions made regarding an ingredient. For example, older actions may not be listed, especially if they do not reflect the agency’s current position.
The Ingredient Directory will be replacing the FDA’s Dietary Ingredient Advisory List, however, anyone signed up to receive updates regarding the advisory list will automatically receive updates to the Ingredient Directory as they become available.
This new directory does not levy any new requirements for dietary supplement manufacturers. What it will do, however, is provide consumers (and manufacturers & retailers), with an easier way to be informed about the types of ingredients found in dietary supplements.
If you would like to submit comments or feedback you can do so by emailing the FDA’s Office of Dietary Supplement Programs.
If you have any questions about the FDA’s new Ingredient Directory or need assistance related to dietary supplement regulations or any other regulatory affairs matters, contact dicentra today.